WORCESTER—A Worcester Polytechnic Institute researcher is joining a major national effort to better understand how a common and often fatal heart condition takes hold—and, ultimately, how it might be stopped.
Kristen Billiar, a professor of biomedical engineering at WPI, will lead part of a $15 million, multi-institution project funded by the American Heart Association focused on early detection and prevention of aortic valve disease. His work centers on a deceptively simple question: why some people with known risk factors develop the disease while others do not.
Billiar has been awarded $1,278,213 over four years to investigate how lipoprotein (a), a cholesterol-carrying particle known as Lp(a), interacts with the physical forces inside the heart to drive calcific aortic valve disease, or CAVD.
CAVD, the most common form of heart valve disease, gradually stiffens and narrows the valve that regulates blood flow from the heart to the rest of the body. The condition contributed to more than 248,000 deaths in the United States in 2019, according to the announcement.
Doctors have long recognized several risk factors, including high blood pressure, inflammation and elevated levels of Lp(a), which is inherited. But the progression from risk to disease remains unclear.
Billiar’s research will examine whether the mechanics of blood flow — including turbulence and the stretching of valve tissue — make heart cells more vulnerable to those circulating risk factors.
“The project at WPI will focus on biomechanics, but we will also collaborate with researchers at other institutions who are concentrating on cell biology and using artificial intelligence (AI) to examine genetic risk factors,” Billiar said in the announcement. “The goal is to identify targets for potential therapies to treat heart valve disorders.”
His work is one of three coordinated projects under the Center for Integrative Valve Science at the University of Pittsburgh, where researchers are combining expertise in engineering, biology and data science to study how the disease develops and progresses.
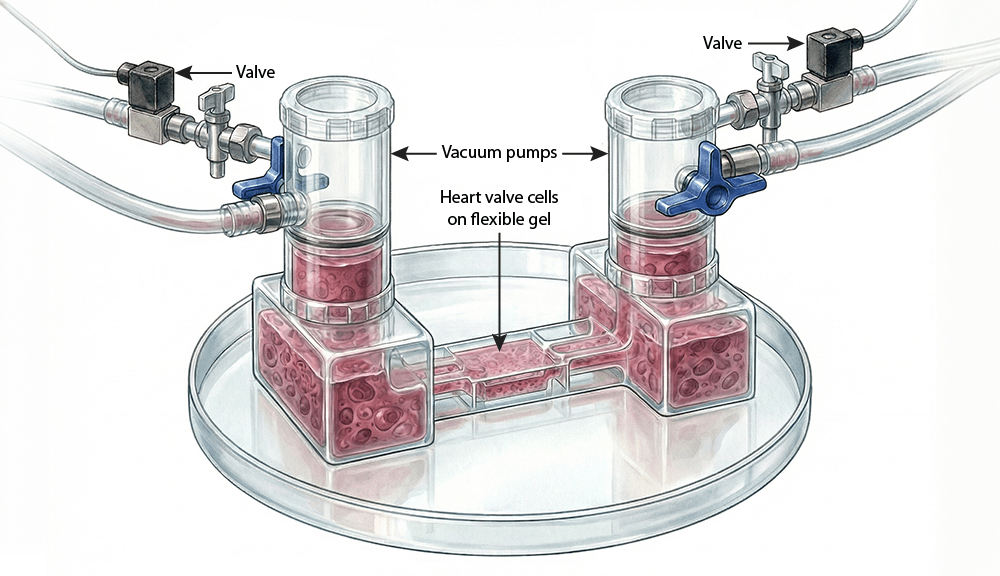
To simulate conditions inside the heart, Billiar’s lab will use “valve-on-a-chip” technology — a system that places human valve cells on flexible materials that can be stretched and exposed to varying patterns of fluid flow.
The approach allows researchers to observe, in controlled conditions, how cells respond to the stresses they would experience inside the body.
“This new project builds on years of research and the development of tools that can reveal the role that mechanical forces play in disease,” Billiar said in the announcement. “Bringing my work together with the work of other researchers has the potential to make great advances in our understanding of heart valve disease and lay the groundwork for new therapeutic approaches.”
The project also involves WPI students, who participate in lab work and research projects tied to the effort.


